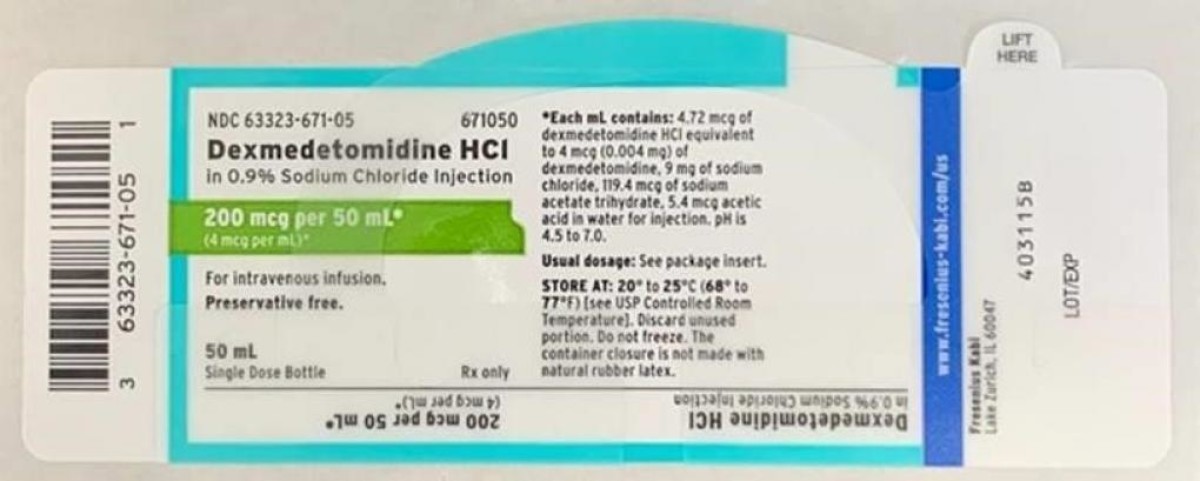
Recall notice
Fresenius Kabi USA Dexmedetomidine HCL 在 0.9% 氯化钠注射液中-由于与利多卡因交叉污染而召回, USA
5 年前 •source fda.gov • 细节
Brand name: Fresenius Kabi USA
Product recalled: Dexmedetomidine HCL in 0.9% Sodium Chloride Injection
Reason of the recall: Cross Contamination with Lidocaine
FDA Recall date: November 19, 2020
Recall details: Fresenius Kabi USA is voluntarily recalling a single lot of Dexmedetomidine HCl…