报告人
重要-您的报告已排队-您的报告可能需要长达 12 小时才能显示在我们的主页上(如果未选择为私人)
访问我们的学习页面了解更多有用的信息,或者发送电子邮件给我们: support@safelyhq.com
Fresenius Kabi USA Dexmedetomidine HCL 在 0.9% 氯化钠注射液中-由于与利多卡因交叉污染而召回, USA
3年前 •source www.fda.gov
Recall notice
United States
Company name: Fresenius Kabi USABrand name: Fresenius Kabi USA
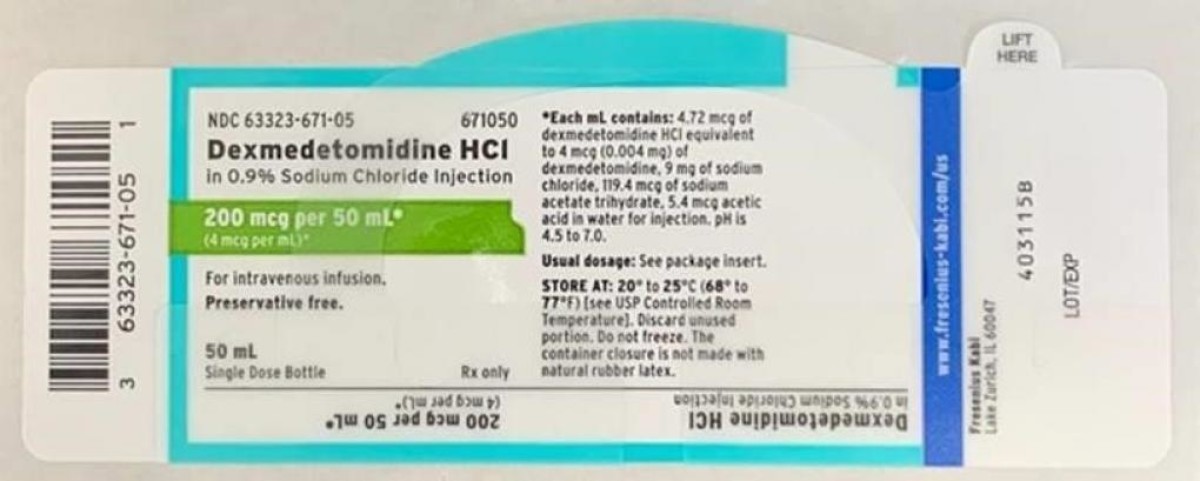
Product recalled: Dexmedetomidine HCL in 0.9% Sodium Chloride Injection
Reason of the recall: Cross Contamination with Lidocaine
FDA Recall date: November 19, 2020
Recall details: Fresenius Kabi USA is voluntarily recalling a single lot of Dexmedetomidine HCl in 0.9% Sodium Chloride Injection, 200 mcg/50 mL (4 mcg /mL), 50 mL fill in a 50 mL vial. Fresenius Kabi initiated this recall due to a trace amount of lidocaine present in the lot. This recall is being performed to the user level. To date, no adverse drug experience reports have been received for the recalled lot. Administration of Dexmedetomidine HCl containing trace amounts of lidocaine to a patient with lidocaine allergy could result in a potentially life-threatening allergic reaction.
Dexmedetomidine Hydrochloride in 0.9% Sodium Chloride Injection is approved for intravenous use and indicated for sedation of non-intubated patients prior to and/or during surgical and other procedures. Listed below is a table of the recalled lot distributed nationwide to wholesalers, distributors, hospitals and pharmacies between April 9, 2020 and April 13, 2020. An image of the label is also included below.
- Dexmedetomidine HCl in 0.9%. Sodium Chloride Injection, 200 mcg / 50 mL (4 mcg / mL), 50 mL fill in a 50 mL vial. NDC Number: 63323-671-50. Product Code: 671050. Batch number: 6123925. Expiration Date: 03/2022. First Ship Date: 04/09/2020. Last Ship Date: 04/13/2020
Fresenius Kabi is notifying its distributors and customers by letter and asking them to check their stock immediately and to quarantine and discontinue the use and distribution of any affected product.
Distributors should notify their customers and direct them to quarantine and discontinue distributing or dispensing any affected lots, and to return the product to Fresenius Kabi.
Customers with questions regarding this recall may contact Fresenius Kabi at 1-866-716-2459 Monday through Friday, during the hours of 8:00 a.m. to 5:00 p.m. Central Time. Consumers should contact their physician or health care provider if they have experienced any problems that may be related to taking or using this drug product.
Check the full recall details on www.fda.gov
Source: FDA
最近有趣的报告
广告故障, Columbus, OH, USA
3周前 •reported by user-dbyfd382
无序的 Subgenix, Rocky Mount, NC, USA
2周前 •reported by user-rkvm7377
未订购的裸露天然生物核糖, Amarillo, TX, USA
4周前 •reported by user-mrhd1868
未经授权的购买, Minnesota, USA
2周前 •reported by user-xnkh2797
我报告了一个未订购的包裹, Alabama, USA
2周前 •reported by user-wbbwf261
我没有花 180 美元订购这样的东西, Romania
1周前 •reported by user-kxjrp853
收到的包裹-二手。胸罩-没有订购, Mechanicsville, VA, USA
1周前 •reported by user-mnqcp927
我没有订购, Oregon, OH, USA
1周前 •reported by user-yxpqq462
我收到了这罐 1 盎司的 Subgenix Bioribose, Xenia, OH, USA
2周前 •reported by user-rbjk6511