Recall notice
Ascend Laboratories Dabigatran Etexilate Capsules - recalled due to Detection of N-Nitrosodimethylamine Impurity, USA
2 years ago •source fda.gov
United States
Ascend Laboratories LLC. is voluntarily recalling Dabigatran Etexilate Capsules. USP 75 mg and 150 mg to the consumer/user level due to the presence of a nitrosamine. N-nitroso-dabigatran, above the established Acceptable Daily Intake (ADI) level. The product lots were distributed nationwide to wholesalers, Distributors and Retailers (dispensers) in the United States from June 2022 to October 2022. To date, Ascend Laboratories LLC., has not received any reports of adverse events related to this recall.Nitrosamines are common in water and foods, including cured and grilled meats, dairy products and vegetables. Everyone is exposed to some level of nitrosamines. These impurities may increase the risk of cancer if people are exposed to them above acceptable levels over long periods of time. The product is used as an oral anticoagulant to lower the risk of stroke and blood clots.
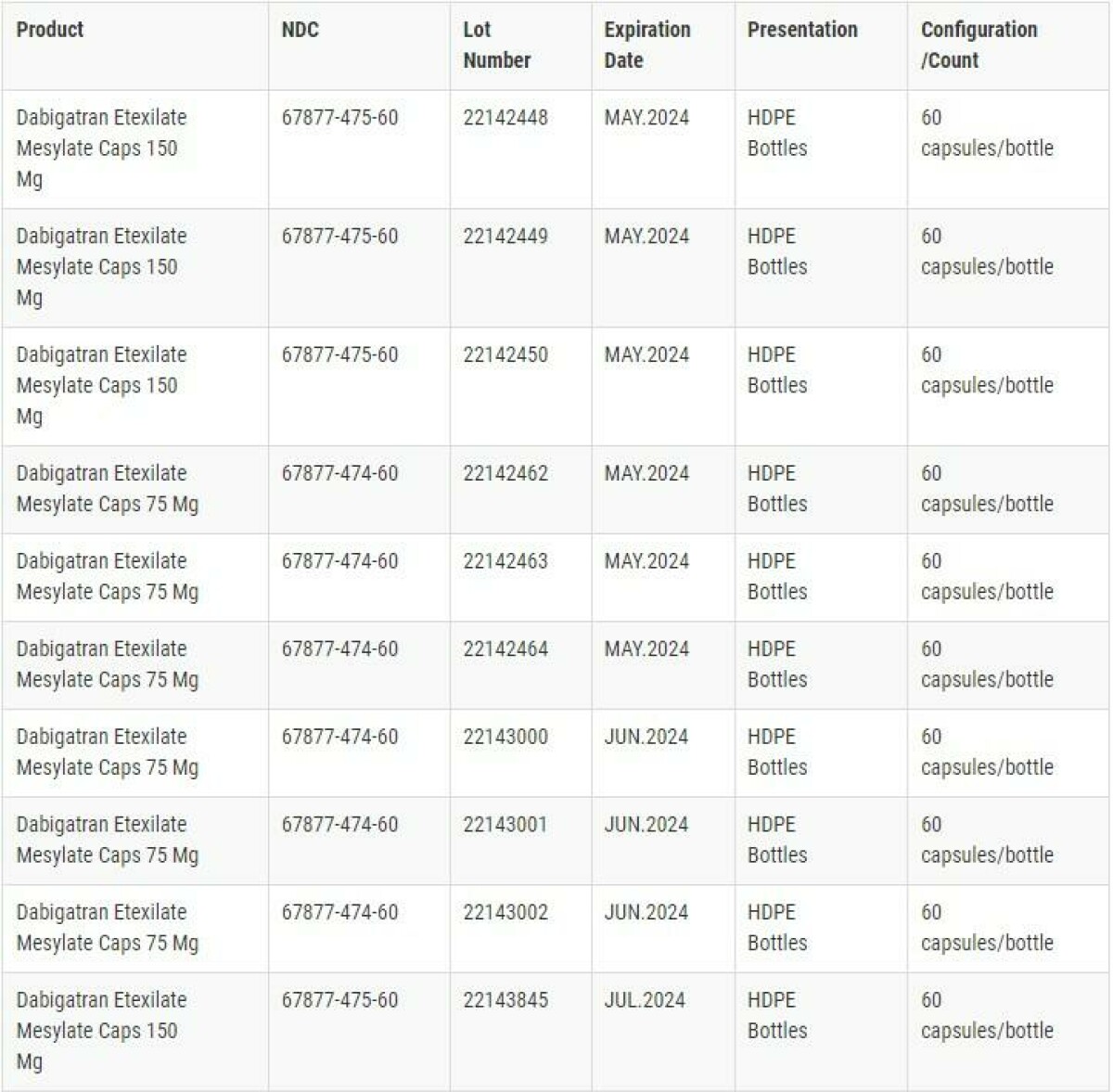
The NDC. Lot Number. Expiration Date. and Packaging Configuration details for Dabigatran Etexilate Capsules that are subjected to this recall are shown in the image below.
Company name: Ascend Laboratories LLC.
Brand name: Ascend Laboratories
Product recalled: Dabigatran Etexilate Capsules, USP
Reason of the recall: Detection of N-Nitrosodimethylamine (NDMA) Impurity
FDA Recall date: March 22, 2023
Source: www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/ascend-laboratories-llc-issues-voluntary-nationwide-recall-dabigatran-etexilate-capsules-usp-75-mg
312
Comments
Comment