Recall notice
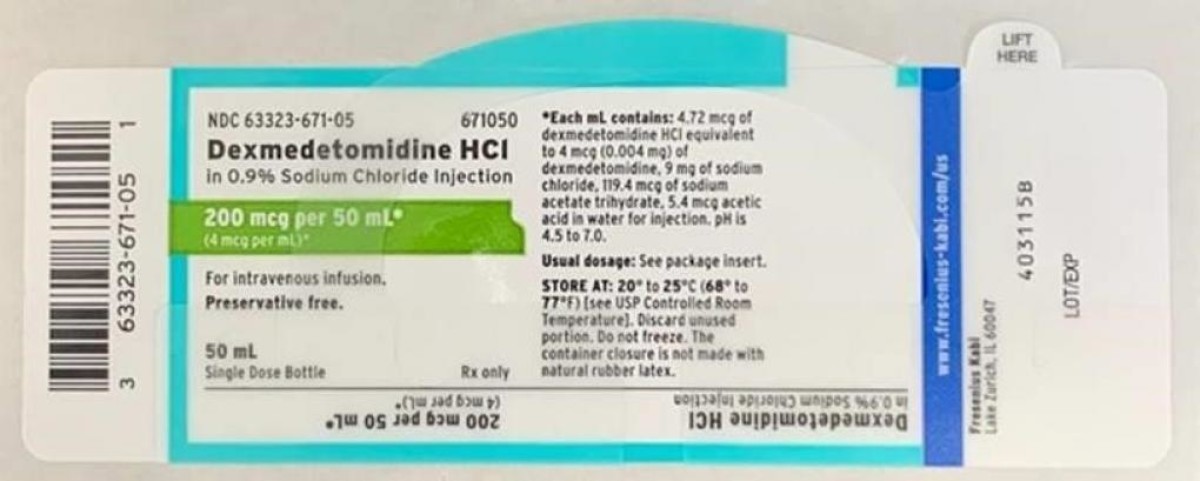
Fresenius Kabi USA Ketorolac Tromethamine Injection, USP, 30 mg/mL - recalled due to Presence of particulate matter, USA
5 years ago •source fda.gov • details
Brand name: Fresenius Kabi
Product recalled: Ketorolac Tromethamine Injection, USP, 30 mg/mL
Reason of the recall: Presence of particulate matter
FDA Recall date: January 08, 2021
Recall details: Fresenius Kabi USA is voluntarily recalling a single lot of Ketorolac Tromethamine Injection, USP,…