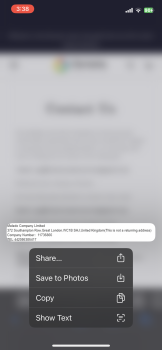
Facebook Scam, 372 Southampton Row, London WC1B 4AE, United Kingdom
3 years ago •reported by user-pccc1746

Southampton Row, England, United Kingdom
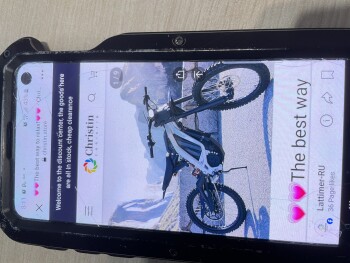
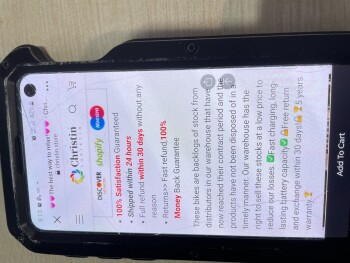
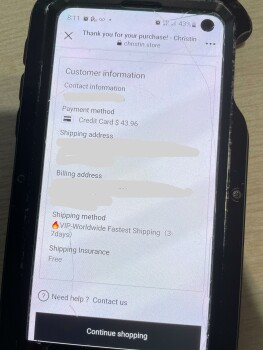
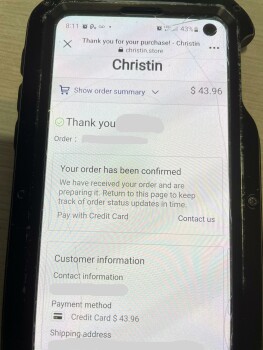
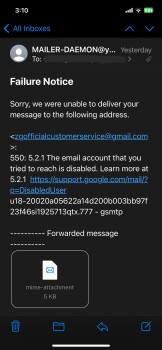
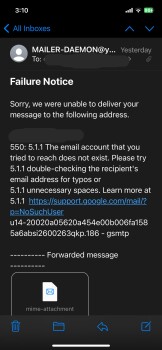
My boyfriend and I was looking on Marketplace(Facebook) then we saw this electric bike for $49.99. There’s a bunch of them who are selling bikes & ebikes for $49.99. We thought it would be helpful if my boyfriend has an electric bike. So checked the webpage and it looks legit to us. Aside from their reason that it is cheap because they are trying to get rid of their stocks, we also love to bike on summer time. And my boyfriend just moved to another city for work reasons as well. Anyways its only $50.00! Who know’s?! So I finished the transaction and after making the payment, I noticed that I didn’t got a confirmation email. Yes, there’s an order no. but that’s all they provided to me. So I went to check if I can contact them and it said they have 24/7 support agents but can be contacted thru email only. So I sent an email but my email went back saying “FAILURE NOTICE”. I thought I made a mistake cuz I just copy pasted it so I double check the email address they have and sent another email and again, my email went back again. Then I look for a way to cancel my order and they don’t have a page to that. Instead, you have to send them an email again but this email address is different from what they are using for support. And like the first two emails I sent, this cancellation email I sent them went back again. I checked my bank account and yes, they already got my money. Today, I went back to their page to see if they have a legit contact info and that’s when I saw their address. But I noticed its says “(This is not the returning address). So I tried to copy paste it again(it’s too long to memorize) and it came out out like its a photo they just edited and made it look like its legit.I'm in Edmonton
506
Comments
Comment