Ausbruch
FDA Advisory Not to Use Addall Dietary Supplements due to Unlawful and Undeclared Ingredients, United States
vor 3 Tagen •source fda.gov
United States
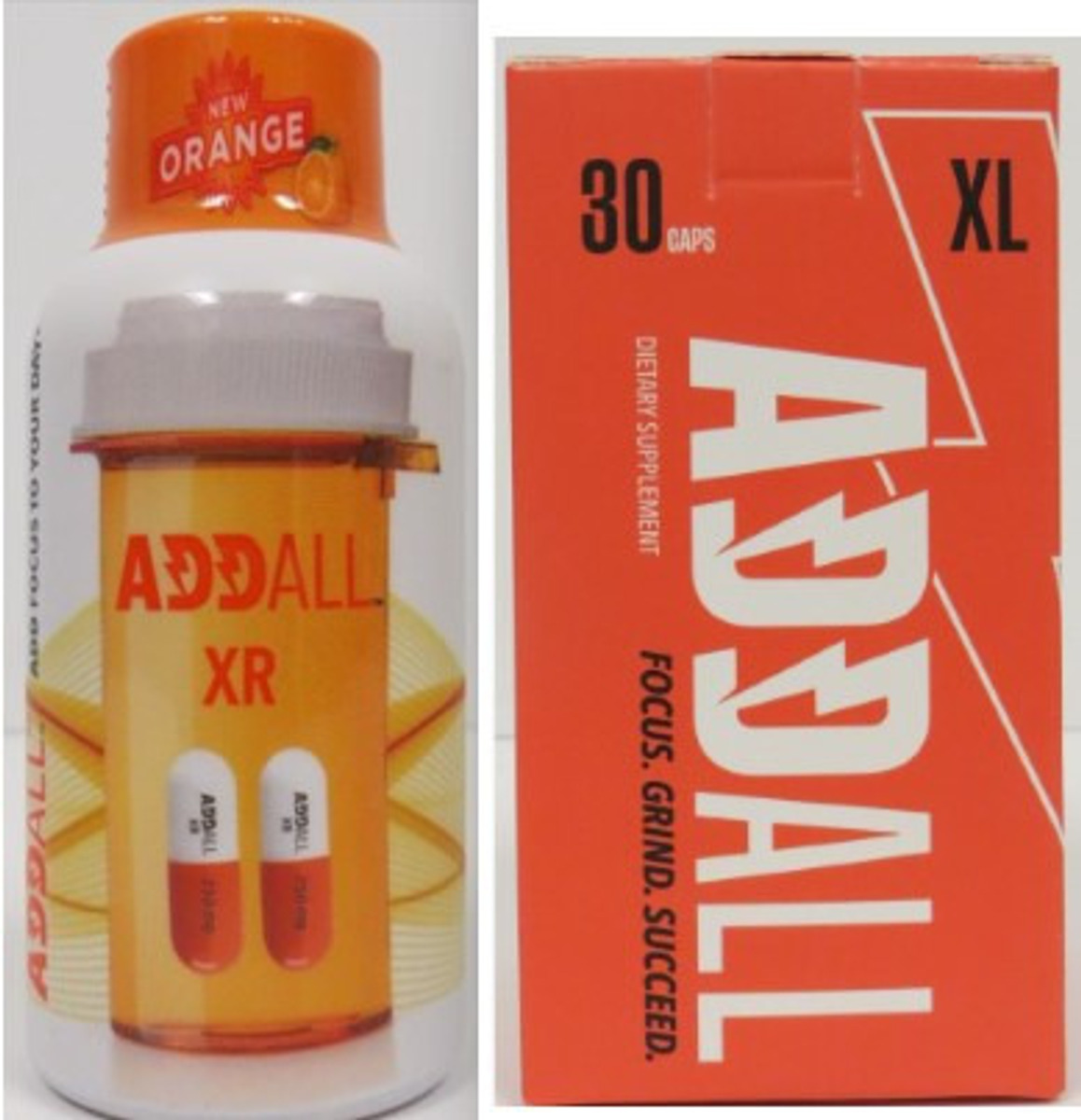
ZMB Enterprises, LLC has issued a recall for its orange-flavored Addall XR Shot and Addall XL capsules due to the presence of unlawful and undeclared ingredients. These dietary supplements, sold online and at retail locations nationwide in the United States, pose serious health risks.The recalled products include:
- The Addall XR Shot, which contains phenibut and undeclared 1,4-DMAA
- The Addall XL capsules, which contain DMHA and undeclared 1,4-DMAA.
The recall affects all sizes and batches of these products.
The issue was discovered through FDA testing, which revealed the presence of these ingredients. The recall was announced on January 6, 2026, when the FDA informed ZMB Enterprises of the findings. The company agreed to recall the Addall XR Shot but declined to recall the Addall XL capsules.
Source: www.fda.gov/food/alerts-advisories-safety-information/fda-advises-consumers-retailers-and-distributors-not-eat-sell-or-distribute-addall-xr-shot-or-addall
Bemerkungen
Kommentar