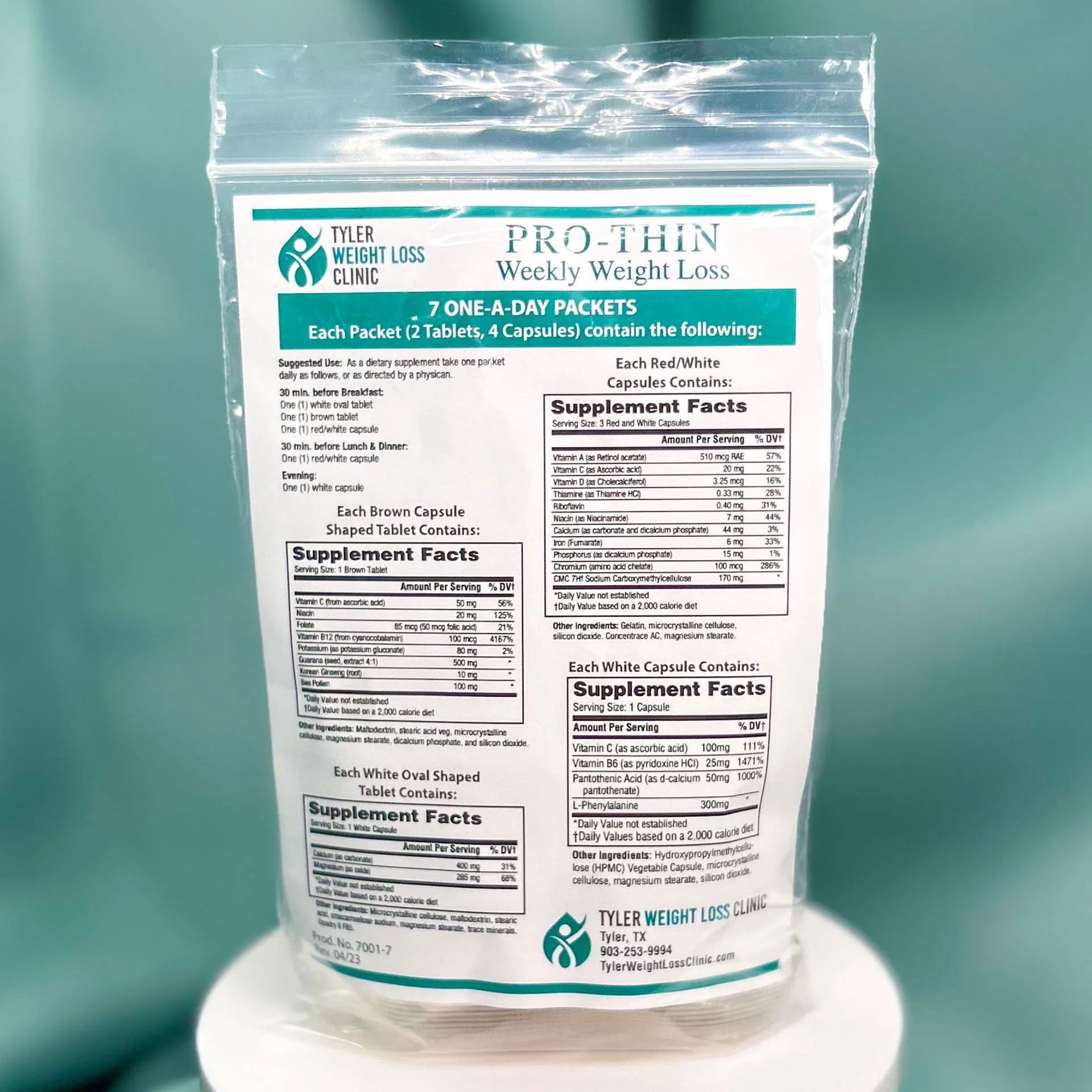
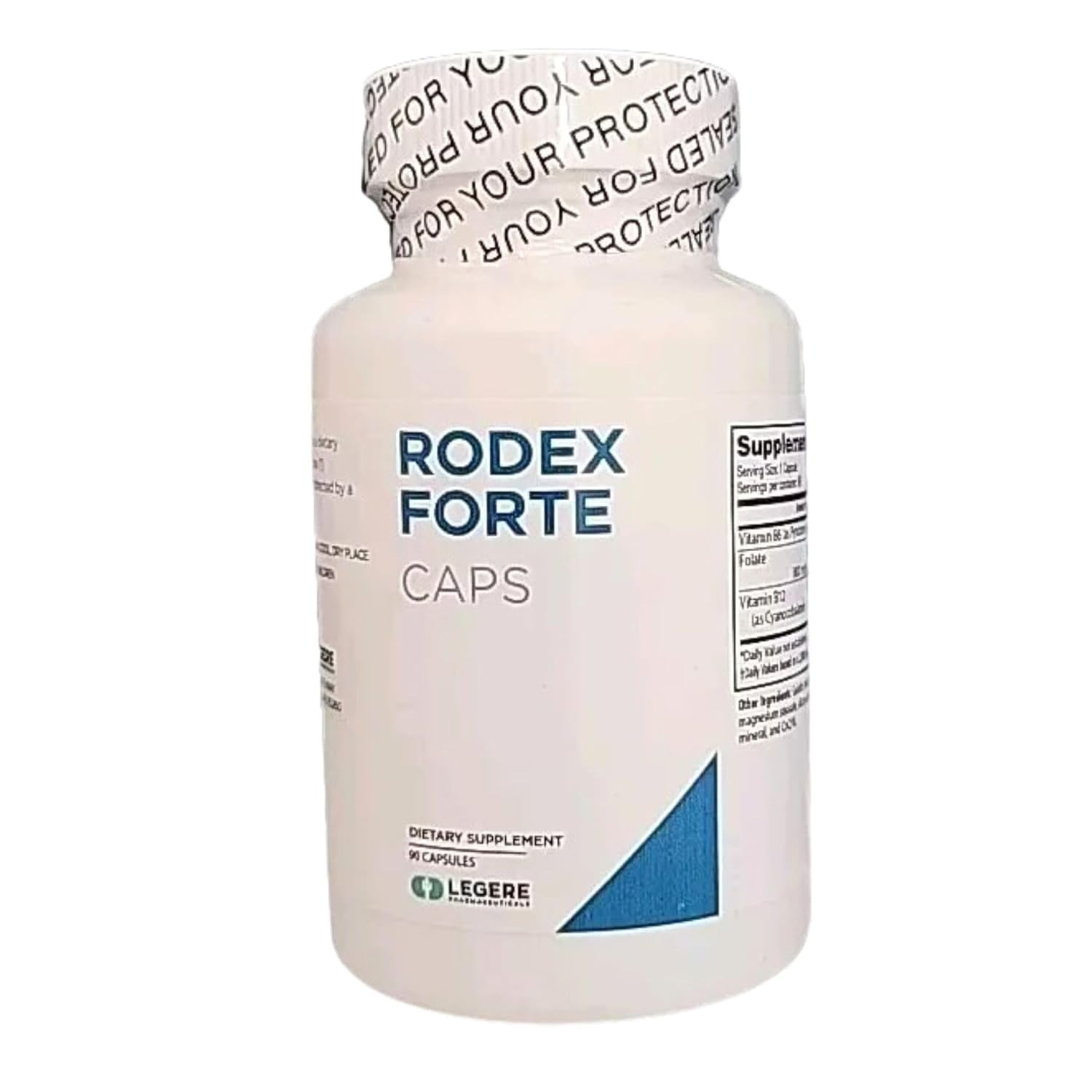
Recall notice
Dwater recalls MyBladder Supplement for bladder support recalled due to E.coli, USA
4 months ago •source fda.gov • details
The recall was classified as Class II.
Source: www.accessdata.fda.gov/scripts/ires/index.cfm
October 28th, 2025:
Purity Products is announcing a recall of one lot of its dietary supplement MyBladder because it has the potential to be contaminated with Escherichia coli O7:K1 and 1303. MyBladder was distributed…