Source: www.wtvm.com
Report by
IMPORTANT - YOUR REPORT IS QUEUED - IT MAY TAKE UP TO 12 HOURS FOR YOUR REPORT TO SHOW ON OUR HOME PAGE (IF NOT OPTED AS PRIVATE)
Visit our learn pages for more helpful information or, email us: support@safelyhq.com
Phenix City, Alabama, United States
Updated:
Source: www.wtvm.com
Recent Interesting Reports
Unordered Package, Santa Rosa, CA, USA
2 days ago •reported by user-pwxmj319
Unordered Supplements, Aldine Westfield Road, Houston, TX, USA
3 weeks ago •reported by user-dtkt5174
I got the wrong shoes, Rossz cipőt kaptam
1 week ago •reported by user-mdwq7578
It's not what I asked, Sulighete, Romania
3 weeks ago •reported by user-xcvy2376
Scam, Monticello, FL, USA
3 weeks ago •reported by user-czvfp313
Scam, Texas, USA
3 weeks ago •reported by user-fnhfd224
Received an item not as described, Montgomery View Drive, Harriman, TN, USA
3 weeks ago •reported by user-vdcpj422
Ring Scam, Yucca Avenue, Lancaster, CA, USA
3 weeks ago •reported by user-rzyzb777
Company on package says YOSHI DEALS
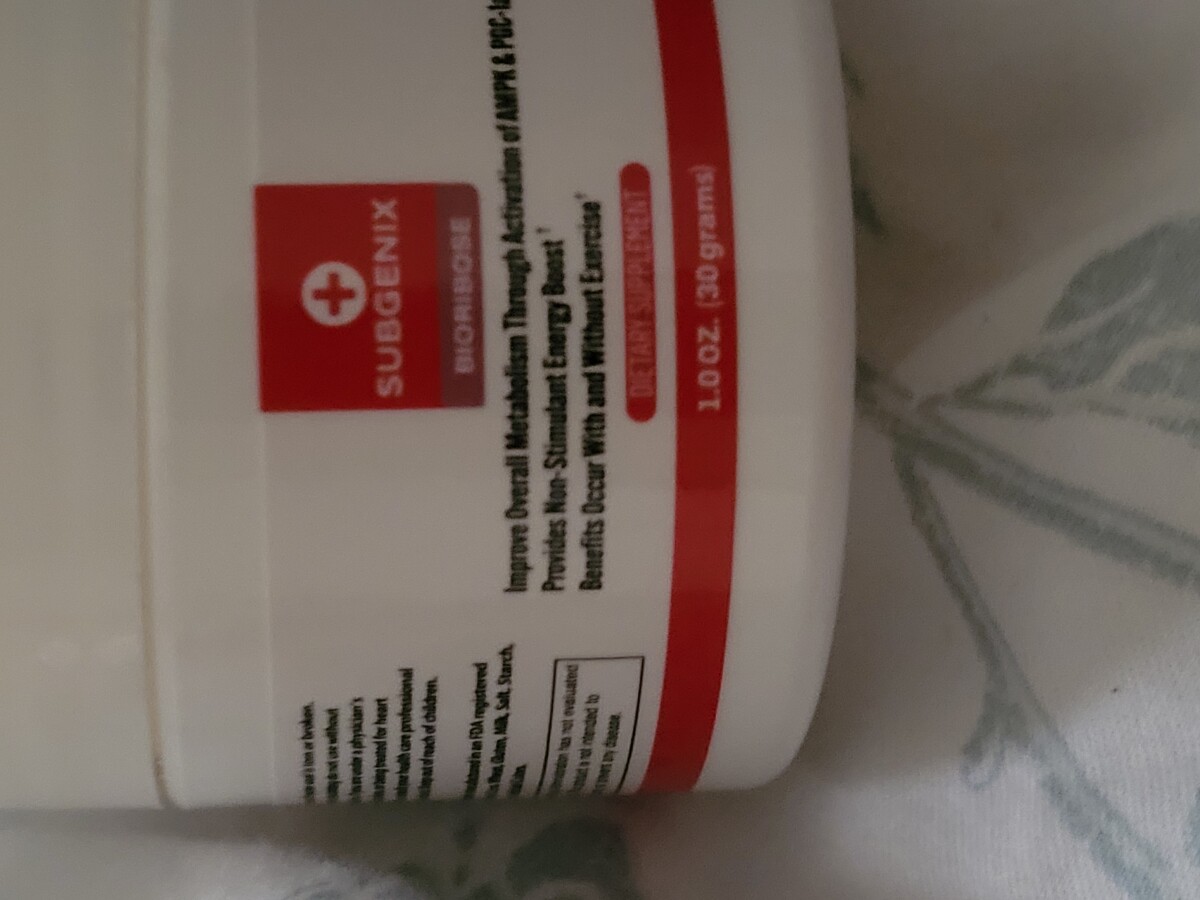
I just received this package also from Tampa Florida... Subgenix, Houston, TX, USA
3 weeks ago •reported by user-fkkg2382
Schwinnng Herbal Dietary Supplement - recalled due to contamination with Nortadalafil, USA
4 days ago •source www.fda.gov
Recall notice
The Schwinnng products contain Nortadalafil, which may interact with nitrates found in some prescription drugs, such as nitroglycerin, and may cause a significant drop in blood pressure that may be life threatening. People with diabetes, high blood pressure, high cholesterol, or heart disease often take nitrates. Among the adult male population, who are most likely to use this product, adult males who use nitrates for cardiac conditions are most at risk.
This product is marketed as a dietary supplement for male sexual enhancement and is packaged and coded as follows:
SCHWINNNG Lot 2108 EXP 10/2024
STOP CLOPEZ CORP is notifying its customers by this press announcement of this recall release and is arranging for a return of all recalled products. Consumers that have a Schwinnng product that is being recalled should stop using and destroy / return if desired to STOP CLOPEZ CORP. Schwinnng products must include the blister pack foil (with any pills remaining) containing all lot codes and receipt of proof of purchase, and your return address.
Consumers should contact their physician or healthcare provider if they have experienced any problems that may be related to taking or using this drug product.
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration. Link to FDA Advisory
If you are experiencing symptoms after eating this product it is important to report it. It can help to detect & resolve outbreaks early and prevent others from being harmed, and it enables better surveillance. If symptoms persist seek medical attention.
Company name: STOP CLOPEZ CORP
Brand name: Schwinnng
Product recalled: Herbal Dietary Supplement
Reason of the recall: Product is tainted with Nortadalafil
FDA Recall date: April 23, 2024
Source: www.fda.gov