Recall notice
Revitaderm, Tridergel Antiseptic Wound Care Gel recalled due to Lysinibacillus fusiformis, USA
vor 1 Woche •source fda.gov
United States
Blaine Labs, Inc. is voluntarily recalling three (3) lot numbers of Wound Care Gel products, 1 oz. & 3 oz. (0.1% Benzalkonium Chloride) to the consumer level due to microbial contamination. Revitaderm® Wound Care Antiseptic Gel 1 oz. & 3 oz. and Tridergel™ Wound Care 1 oz. was distributed nationwide in the U.S to healthcare providers (doctor’s offices) they were not distributed for retail or internet sales.The affected product has been found to contain Lysinibacillus fusiformis, an environmental organism.
Risk Statement:
This voluntary recall has been initiated as a precautionary measure due to the detection of microbial presence. The tested samples identified the presence of Lysinibacillus fusiformis. Lysinibacillus fusiformis is an environmental, spore-forming bacterium.
While it is generally considered to have low pathogenicity in healthy individuals, it can survive in adverse conditions and may act as an opportunistic organism. Patients with open wounds, compromised skin barriers, or weakened immune systems (e.g., elderly, diabetic, or immunocompromised individuals) may be at increased risk for infection, skin irritation, and / or delayed wound healing. In immunocompromised individuals, including those with poorly controlled diabetes, elderly individuals with immune senescence, and young children with an immature immune system, these infections may become severe and lead to life-threatening complications such as endocarditis or central system infection.
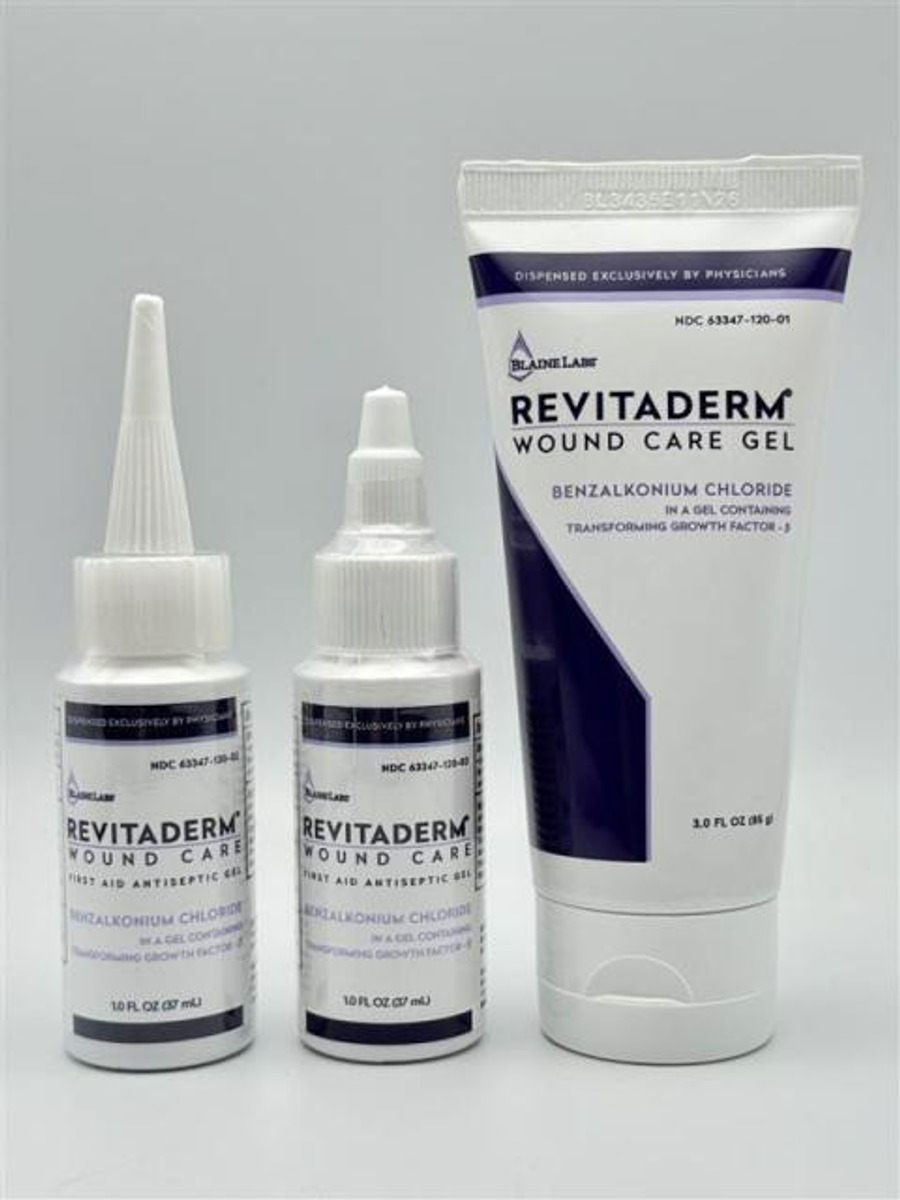
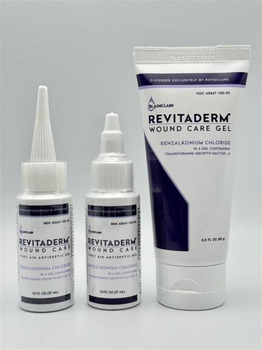
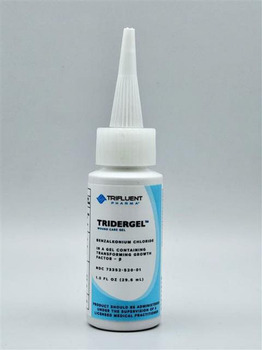
The product is used as a topical antiseptic for first aid to help prevent the risk of infection in minor cuts, scrapes, and burns and is packaged in a 1 oz. bottle and 3 oz. tube. The Revitaderm® Wound Care Gel can be identified by its purple trim and Tridergel™ Wound Care can be identified by its light blue trim. The 1 oz. bottle has two cap configurations, twist and longer pointy cap, and is secured with a shrink / body band. The bottle has the expiration date and lot number printed on the bottom portion of the bottle. The 3 oz. is a tube and contains the lot number and expiration date imprinted along the crimp at the top of the tube.
PRODUCT: 1 oz. & 3 oz. Revitaderm® Wound Care Antiseptic Gel
- Active Ingredient: Benzalkonium Chloride 0.1%
- Lot Number/EXP Date/Packaging: BL3608, 07/01/28 Printed with Yellow ink on the bottom of the bottle (1 oz.), shorter twist cap.
- Lot Number/EXP Date/Packaging: BL3435, 11/06/26 Printed with Black ink on the bottom of the bottle (1 oz.), long pointy cap
- Lot Number/EXP Date/Packaging: BL3435, 11/06/26 Imprinted on the tube crimp (3 oz.)
- Lot Number/EXP Date/Packaging: BL3525, 08/07/27 Printed with Black ink on the bottom of the bottle (1 oz.), long pointy cap
- Lot Number/EXP Date/Packaging: BL3525, 08/07/27 Imprinted on the tube crimp (3 oz.)
PRODUCT: 1 oz. Tridergel™ Wound Care
- Active Ingredient: Benzalkonium Chloride 0.1%
- Lot Number/EXP Date/Packaging: BL3435, 11/06/26 Printed with Black ink on the bottom of the bottle (1 oz.), long pointy cap
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.
Source: www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/blaine-labs-inc-issues-voluntary-nationwide-recall-wound-care-gel-products-due-microbial
27
Bemerkungen
Kommentar