Recall notice
Tuymec Minoxidil Hair Growth Spray Bottles recalled due to Packaging Issue, United States
il y a 1 semaine •source cpsc.gov
United States
DrHealBeauty has issued a recall for Tuymec Minoxidil Hair Growth Kits due to non-compliance with child-resistant packaging standards, posing a poisoning risk to children. The product was sold online on Amazon from June to August 2025.HAZARD:
The hair serum includes minoxidil, an ingredient that is required under the Poison Prevention Packaging Act to be sold in child-resistant packaging. However, this product is not packaged in a child-resistant container, which creates a risk of serious harm or poisoning if young children accidentally ingest it.
AFFECTED PRODUCT
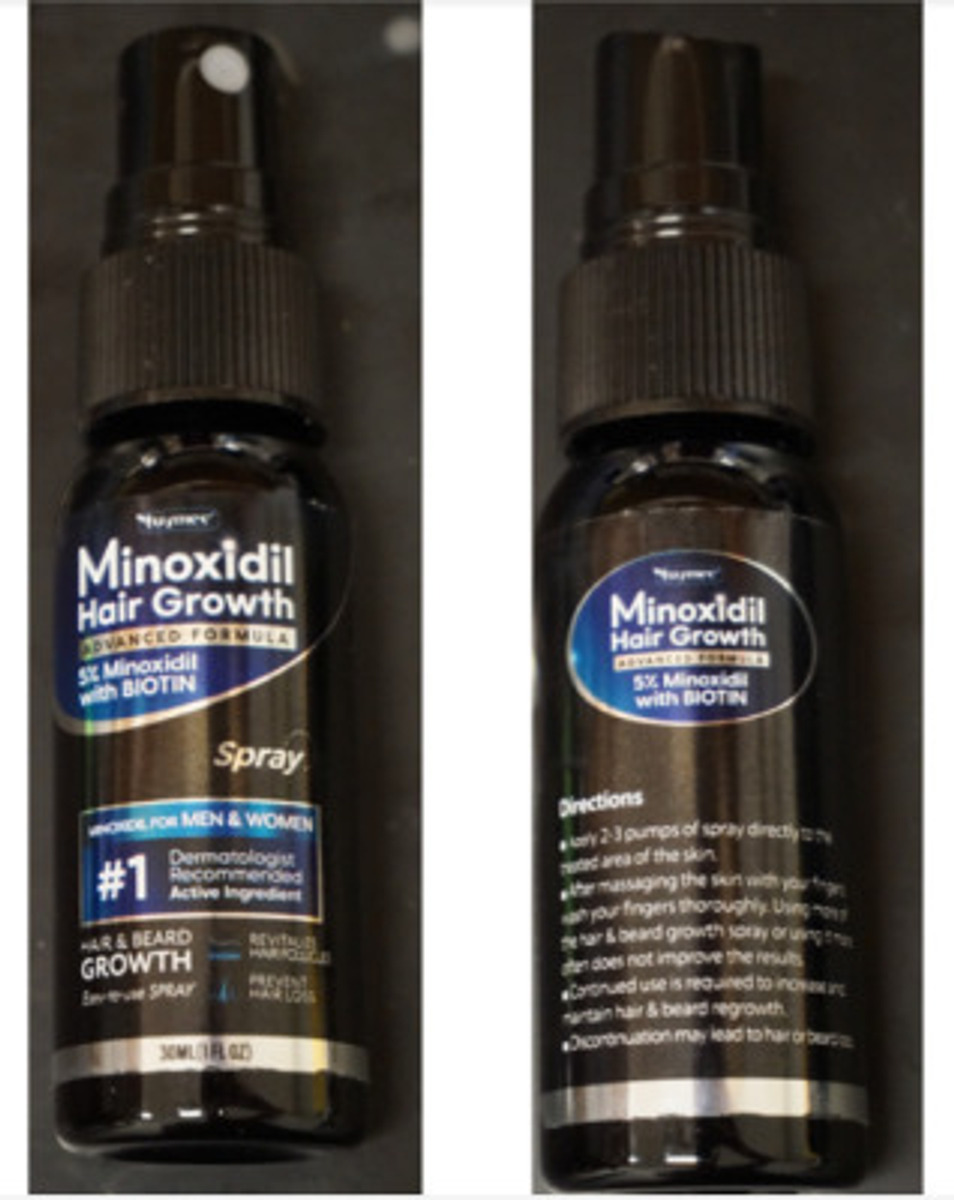
PRODUCT: Tuymec Minoxidil Hair Growth Kits, 5% Minoxidil with Biotin Spray
- Size: Kit with two spray bottles
- Two black spray bottles labeled:
“Tuymec Minoxidil Hair Growth”
“Advanced Formula”
“5% Minoxidil with BIOTIN”
- Packaged in a black carton with white and silver lettering
- Includes “Drug Facts,” distributor details, product claim icons, and batch/manufacture/expiration dates
- Sold online at Amazon.com (June–August 2025)
- About 6,200 units affected
The issue was identified due to the packaging not meeting the Poison Prevention Packaging Act requirements. The recall was announced on April 2, 2026.
Source: www.cpsc.gov/Recalls/2026/Tuymec-Minoxidil-Hair-Growth-Spray-Bottles-Recalled-Due-to-Risk-of-Serious-Injury-or-Death-from-Child-Poisoning-Violates-Mandatory-Standard-for-Child-Resistant-Packaging-Sold-on-Amazon-by-DrHealBeauty
27
Commentaires
Commentaire