Recall notice
Top Care Eye Drops recalled due to Issues with the Manufacturer's Aseptic Filling Process, United States
4 weeks ago •source webseal.topsmarkets.com
United States
TOPS Markets has issued a recall for several Top Care Eye Drops products due to potential concerns with the manufacturer's aseptic filling process. The affected products were sold at TOPS locations (New York, Pennsylvania, and Vermont).AFFECTED PRODUCTS:
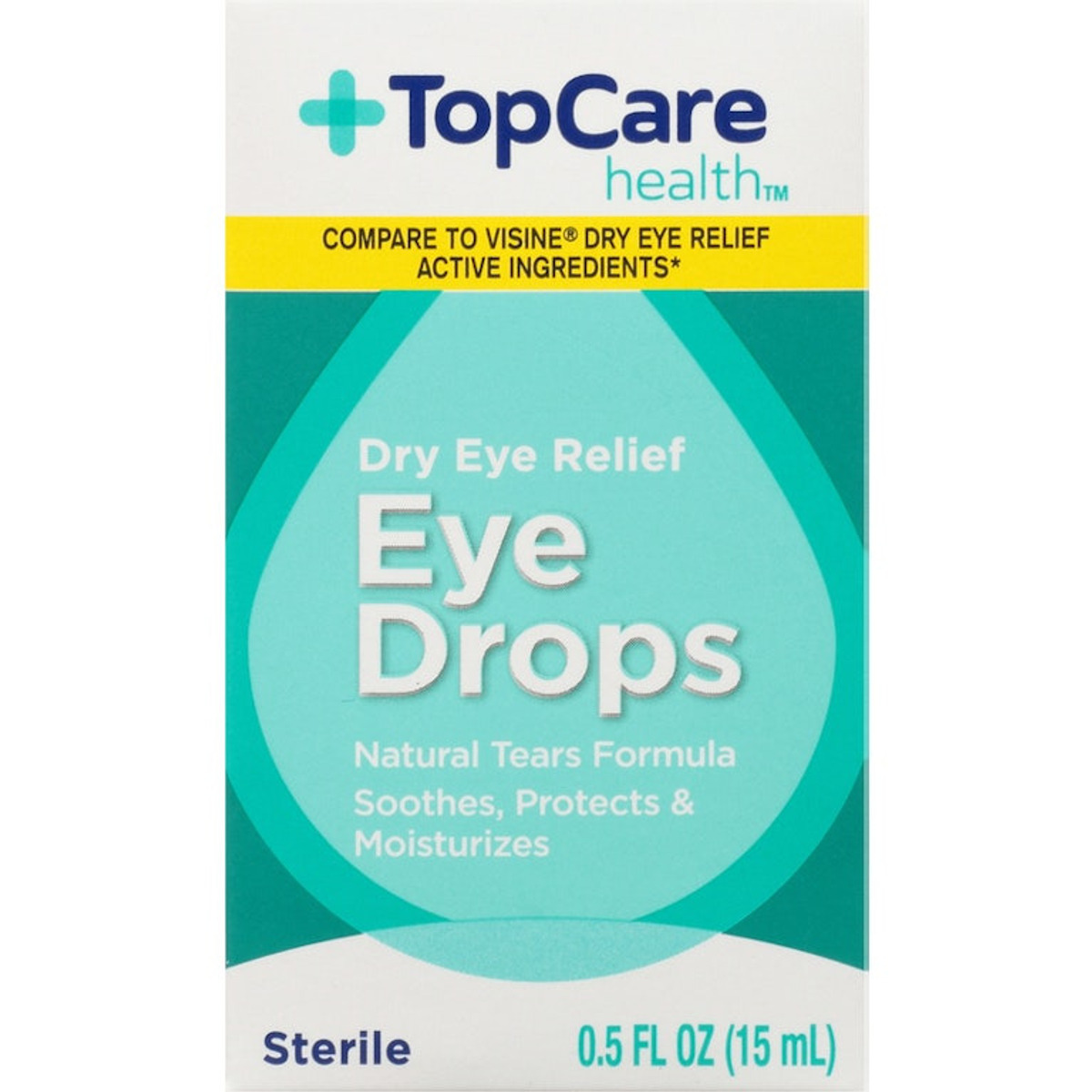
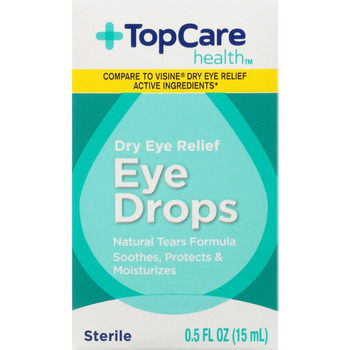
PRODUCT: Product: TOP CARE EYE DROPS DRY RELIEF
- Code Date: All
- Size: .50 OZ
- UPC: 00003680036100
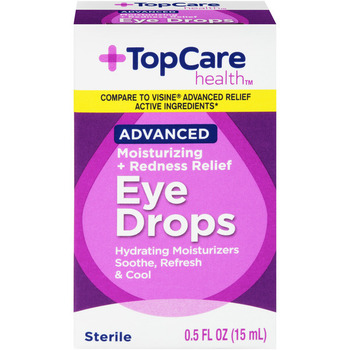
PRODUCT: TOP CARE EYE DROPS ADV MOISTUR
- Code Date: All
- Size: .50 FOZ
- UPC: 00003680003641
PRODUCT: TOP CARE ARTIFCL TEARS EYE DRP
- Code Date: ALL
- Size: .50 FOZ
- UPC: 00003680009331
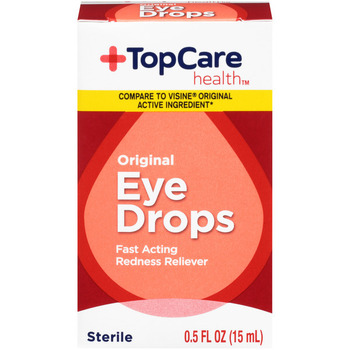
PRODUCT: TOP CARE EYE DROPS ORIGINAL
- Code Date: ALL
- Size: .50 FOZ
- UPC: 00003680003639
The recall was announced on March 6, 2026, and is classified as a Class I recall. This classification indicates a serious health risk.
Source: webseal.topsmarkets.com/jwww2/www2/ProdRecall/index.cfm
Comments
Comment