Source: Osha.gov | Receipt Date: 2020-11-19
Cross Contamination
Updated:
Source: Osha.gov | Receipt Date: 2020-11-19
Fresenius Kabi USA Dexmedetomidine HCL in 0.9% Sodium Chloride Injection - recalled due to cross contamination with Lidocaine, USA
3 years ago •source www.fda.gov
Recall notice
Brand name: Fresenius Kabi USA
Product recalled: Dexmedetomidine HCL in 0.9% Sodium Chloride Injection
Reason of the recall: Cross Contamination with Lidocaine
FDA Recall date: November 19, 2020
Recall details: Fresenius Kabi USA is voluntarily recalling a single lot of Dexmedetomidine HCl …
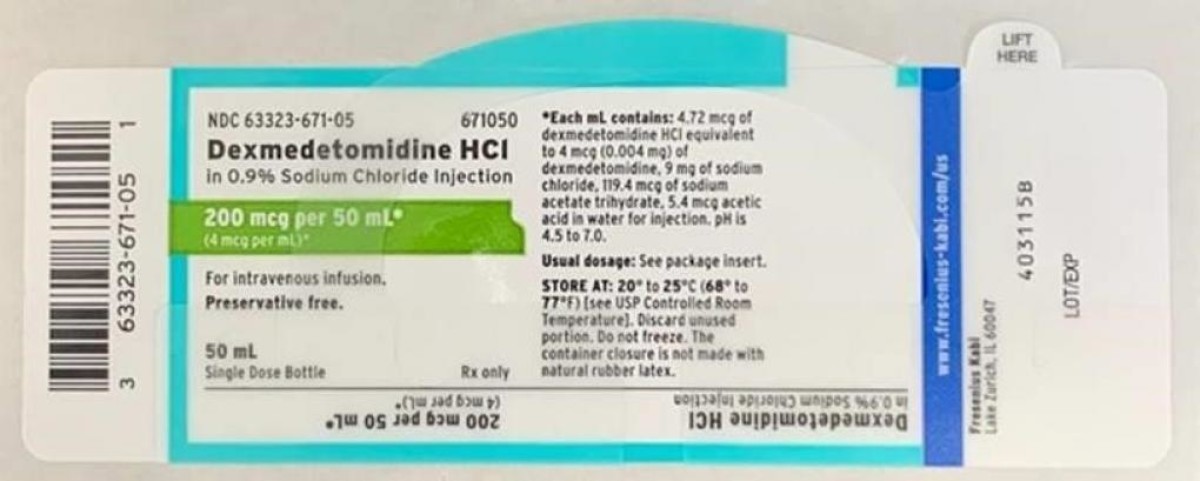
Dexmedetomidine Hydrochloride in 0.9% Sodium Chloride Injection is approved for intravenous use and indicated for sedation of non-intubated patients prior to and/or during surgical and other procedures. Listed below is a table of the recalled lot distributed nationwide to wholesalers, distributors, hospitals and pharmacies between April 9, 2020 and April 13, 2020. An image of the label is also included below.
- Dexmedetomidine HCl in 0.9%. Sodium Chloride Injection, 200 mcg / 50 mL (4 mcg / mL), 50 mL fill in a 50 mL vial. NDC Number: 63323-671-50. Product Code: 671050. Batch number: 6123925. Expiration Date: 03/2022. First Ship Date: 04/09/2020. Last Ship Date: 04/13/2020
Fresenius Kabi is notifying its distributors and customers by letter and asking them to check their stock immediately and to quarantine and discontinue the use and distribution of any affected product.
Distributors should notify their customers and direct them to quarantine and discontinue distributing or dispensing any affected lots, and to return the product to Fresenius Kabi.
Customers with questions regarding this recall may contact Fresenius Kabi at 1-866-716-2459 Monday through Friday, during the hours of 8:00 a.m. to 5:00 p.m. Central Time. Consumers should contact their physician or health care provider if they have experienced any problems that may be related to taking or using this drug product.
Check the full recall details on www.fda.gov
Source: FDA
Recent Interesting Reports
Subgenix Bioribose, Washington D.C., DC, USA
4 days ago •reported by user-nfgyz444
Package I did not order, Atlanta, GA, USA
4 weeks ago •reported by user-rvbt9364
I didn't order a smart watch, Walton Street, Durham, NC, USA
2 weeks ago •reported by user-xjtyj214
This company is a fake I didn't receive the box that was advertised
received an unsolicited package of Subgenix-bioribose, Warwick, RI, USA
2 weeks ago •reported by user-kpvbj826
I received a container of Subgenix that I didn't order., Oakhurst, CA, USA
6 days ago •reported by user-ktyv7427
get my address??
Did not order, Walnut, IA, USA
2 days ago •reported by user-rxnr3346
Unordered gift from China, Ettalong Beach NSW, Australia
2 weeks ago •reported by user-mywn5516
Li yangling.
From Jiutangda Logistic Park.
ChangShou
3rd zhongxiang. Ji Jin men Hube
China
Did not order the gift have packaged it back up and sent it back to China.
Scam Alert, 2207 Summit Ave, Union City, NJ, USA
4 weeks ago •reported by user-hczdm183
Rival Smartwatch. (Info @orders4. me), New Paris, OH, USA
4 weeks ago •reported by user-kjny1173
I received a package I did not order, Tucson, AZ, USA
1 week ago •reported by user-fwtc8752
from 2800 N Franklin Rd, Indianapolis, IN 46329
Report by
IMPORTANT - YOUR REPORT IS QUEUED - IT MAY TAKE UP TO 12 HOURS FOR YOUR REPORT TO SHOW ON OUR HOME PAGE (IF NOT OPTED AS PRIVATE)
Visit our learn pages for more helpful information or, email us: support@safelyhq.com