Source: Osha.gov | Receipt Date: 2020-11-19
Cross Contamination
Updated:
Source: Osha.gov | Receipt Date: 2020-11-19
Fresenius Kabi USA Dexmedetomidine HCL in 0.9% Sodium Chloride Injection - recalled due to cross contamination with Lidocaine, USA
3 years ago •source www.fda.gov
Recall notice
Brand name: Fresenius Kabi USA
Product recalled: Dexmedetomidine HCL in 0.9% Sodium Chloride Injection
Reason of the recall: Cross Contamination with Lidocaine
FDA Recall date: November 19, 2020
Recall details: Fresenius Kabi USA is voluntarily recalling a single lot of Dexmedetomidine HCl …
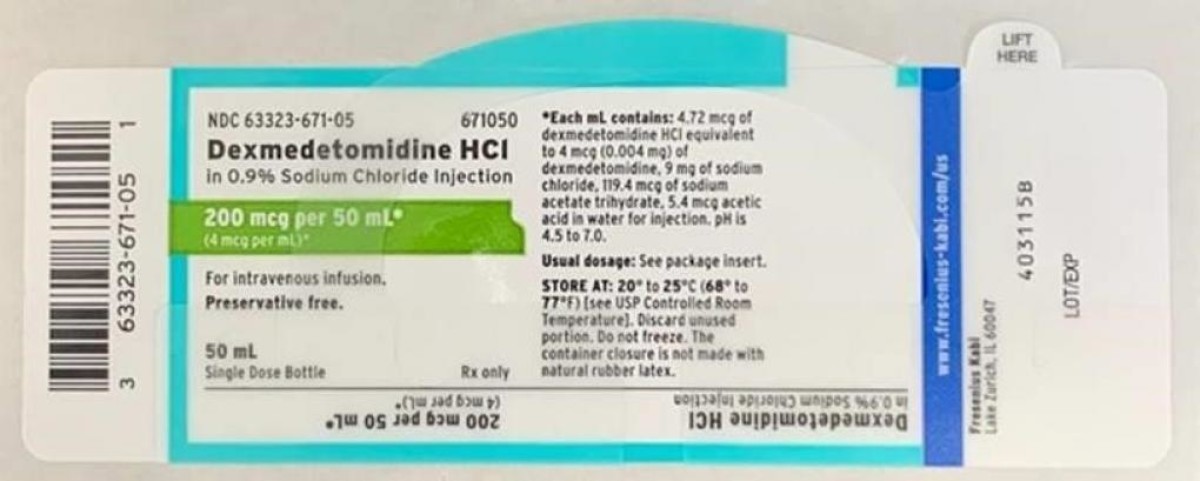
Dexmedetomidine Hydrochloride in 0.9% Sodium Chloride Injection is approved for intravenous use and indicated for sedation of non-intubated patients prior to and/or during surgical and other procedures. Listed below is a table of the recalled lot distributed nationwide to wholesalers, distributors, hospitals and pharmacies between April 9, 2020 and April 13, 2020. An image of the label is also included below.
- Dexmedetomidine HCl in 0.9%. Sodium Chloride Injection, 200 mcg / 50 mL (4 mcg / mL), 50 mL fill in a 50 mL vial. NDC Number: 63323-671-50. Product Code: 671050. Batch number: 6123925. Expiration Date: 03/2022. First Ship Date: 04/09/2020. Last Ship Date: 04/13/2020
Fresenius Kabi is notifying its distributors and customers by letter and asking them to check their stock immediately and to quarantine and discontinue the use and distribution of any affected product.
Distributors should notify their customers and direct them to quarantine and discontinue distributing or dispensing any affected lots, and to return the product to Fresenius Kabi.
Customers with questions regarding this recall may contact Fresenius Kabi at 1-866-716-2459 Monday through Friday, during the hours of 8:00 a.m. to 5:00 p.m. Central Time. Consumers should contact their physician or health care provider if they have experienced any problems that may be related to taking or using this drug product.
Check the full recall details on www.fda.gov
Source: FDA
Recent Interesting Reports
A ring I did not order from GRA, Michigan, USA
3 weeks ago •reported by user-wrvfy174
44211 Yucca Ave.,Lancaster CA 93534
GRA moissanite
GRA 89007****
Scam, Salem, IN, USA
3 weeks ago •reported by user-fpprv682
Small bottle in plastic mailing envelope. Return address is:
Fulfillment House
PO Box 5708
Tampa, Fl 33675-5708
The Package came from DP 192-01 Northern BLVD Flushing NY 11358 I did not order this package
The package came from
Online scam, Fort Pierce, FL, USA
2 weeks ago •reported by user-pxyrp196
Unwanted purchases, 2207 Summit Avenue, Union City, NJ 07087, USA
3 weeks ago •reported by user-pgjrm872
Ring sent I did not order from California, From California im in tucson
1 week ago •reported by user-tkvjf322
Fraud scam, Terre Haute, Indiana 47802, USA
3 weeks ago •reported by user-wxzf1878
Received an item not as described, Montgomery View Drive, Harriman, TN, USA
2 weeks ago •reported by user-vdcpj422
Shoes not received yet, Georgia, USA
5 days ago •reported by user-zjjcp163
I received a package for a small jar of Keto pure I have no idea why this came, Tampa, FL, USA
5 days ago •reported by user-yryy6943
Report by
IMPORTANT - YOUR REPORT IS QUEUED - IT MAY TAKE UP TO 12 HOURS FOR YOUR REPORT TO SHOW ON OUR HOME PAGE (IF NOT OPTED AS PRIVATE)
Visit our learn pages for more helpful information or, email us: support@safelyhq.com