Source: Osha.gov | Receipt Date: 2020-11-19
Cross Contamination
Updated:
Source: Osha.gov | Receipt Date: 2020-11-19
Fresenius Kabi USA Dexmedetomidine HCL in 0.9% Sodium Chloride Injection - recalled due to cross contamination with Lidocaine, USA
3 years ago •source www.fda.gov
Recall notice
Brand name: Fresenius Kabi USA
Product recalled: Dexmedetomidine HCL in 0.9% Sodium Chloride Injection
Reason of the recall: Cross Contamination with Lidocaine
FDA Recall date: November 19, 2020
Recall details: Fresenius Kabi USA is voluntarily recalling a single lot of Dexmedetomidine HCl …
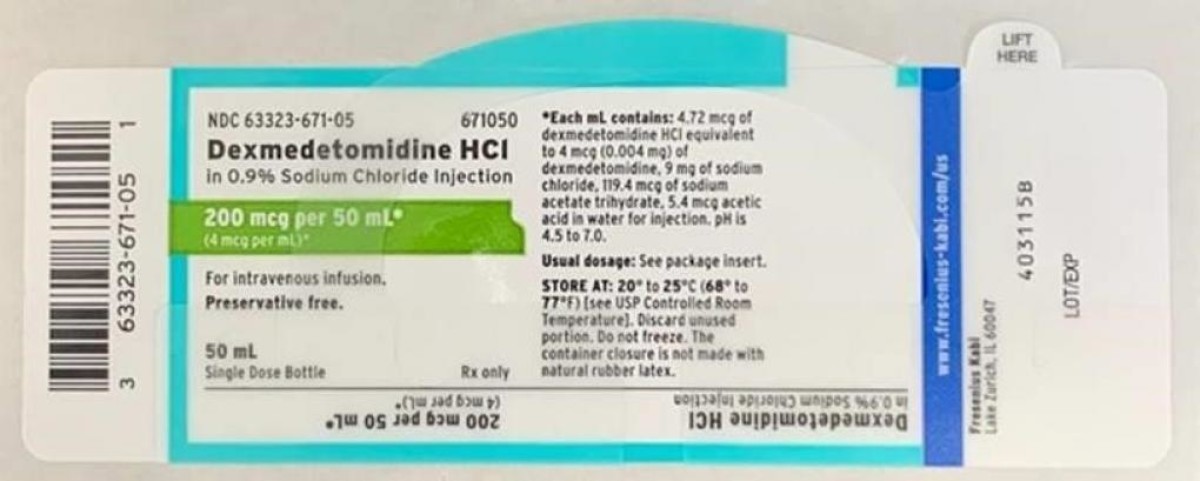
Dexmedetomidine Hydrochloride in 0.9% Sodium Chloride Injection is approved for intravenous use and indicated for sedation of non-intubated patients prior to and/or during surgical and other procedures. Listed below is a table of the recalled lot distributed nationwide to wholesalers, distributors, hospitals and pharmacies between April 9, 2020 and April 13, 2020. An image of the label is also included below.
- Dexmedetomidine HCl in 0.9%. Sodium Chloride Injection, 200 mcg / 50 mL (4 mcg / mL), 50 mL fill in a 50 mL vial. NDC Number: 63323-671-50. Product Code: 671050. Batch number: 6123925. Expiration Date: 03/2022. First Ship Date: 04/09/2020. Last Ship Date: 04/13/2020
Fresenius Kabi is notifying its distributors and customers by letter and asking them to check their stock immediately and to quarantine and discontinue the use and distribution of any affected product.
Distributors should notify their customers and direct them to quarantine and discontinue distributing or dispensing any affected lots, and to return the product to Fresenius Kabi.
Customers with questions regarding this recall may contact Fresenius Kabi at 1-866-716-2459 Monday through Friday, during the hours of 8:00 a.m. to 5:00 p.m. Central Time. Consumers should contact their physician or health care provider if they have experienced any problems that may be related to taking or using this drug product.
Check the full recall details on www.fda.gov
Source: FDA
Recent Interesting Reports
Second delivery I didn’t order., Atlanta, GA, USA
2 weeks ago •reported by user-ddgbb264
I ordered a citizen watch and got another watch, București, Romania
2 days ago •reported by user-cvqnh935
Package whitout being ask, Am Eifeltor 501, 50997 Köln, Rodenkirchen, Germany
3 weeks ago •reported by user-brqd6771
Return DI RETURN ONLY, NOT FOR SALES Postlink Am Eifeltor 501, 50997 Köln, Germany
I received a package from heather mottys, Hudsonville, Michigan, USA
2 weeks ago •reported by user-bjwg9873
That does not look at all what I ordered. How do I return and get a refund
A product I did not order from Verti Products from Tampa, FL, Yorba Linda, CA, USA
4 weeks ago •reported by user-mqqqh719
I received a jar of subjenix bioribose that I did not order., San Francisco, CA, USA
5 days ago •reported by user-qpqwb441
Unsolicited package, Syracuse, NY, USA
4 days ago •reported by user-xnzf4231
Empty Package, Levittown, PA, USA
1 week ago •reported by user-ydpht521
Nutrena Country Feeds Meat bird 22% Crumble (RV) - recalled due to non-inclusion of vitamin D, Bryson City, North Carolina, USA
6 days ago •source www.fda.gov
Recall notice
Lack of Vitamin D in meat bird diets can lead to mineral deficiencies and bone issues, including rickets in growing birds. Symptoms include lameness and rubbery bones.
The recalled product is:
- Product: Nutrena® Country Feeds® Meatbird 22% Crumble (RV), Size: 50 lb. bag, Lot Code: All lots, Product Code: 95188, Species: Meat birds (Broilers, Turkeys, Ducks, Geese and Pheasants, Manufacture dates: July 2022-March 2024, Shelf Life (Days): 120.
The lot code and manufacture date can be found on the bottom right-hand side of the label.
Consumers and other end users who have any of the affected lots in their possession should return remaining product to their local dealer or retailer for a replacement or full refund.
In case your birds experience harm from this product, it is important to report it. It can help to detect & resolve issues and prevent others from being harmed, and it enables better surveillance.
Company name: Cargill
Brand name: Nutrena Country Feeds
Product recalled: Meat bird 22% Crumble (RV)
Reason of the recall: Non-inclusion of Vitamin D
FDA Recall date: April 13, 2024
Source: www.fda.gov
I ordered from this and I never received the merchandise i order since March 24.2024, Texas, USA
1 week ago •reported by user-dncf2793
Report by
IMPORTANT - YOUR REPORT IS QUEUED - IT MAY TAKE UP TO 12 HOURS FOR YOUR REPORT TO SHOW ON OUR HOME PAGE (IF NOT OPTED AS PRIVATE)
Visit our learn pages for more helpful information or, email us: support@safelyhq.com